#fluids
Stone Ocean Villain Run 4: [White Snake]
Say your sweetest prayer and hope the right God answers you
White Snake follows the main villain tradition of being very very hot
Post link
Figs. 11 & 12. “Mouvement d’un fluide dans un plan.” Archives des sciences physiques et naturelles. 1898.
Post link
Flow galleria (#2) : Smoke from an Incense stick
In this set of images the smoke from an incense stick is seen making a transition from a smooth laminar flow (very close to the incense stick) to a turbulent flow (far away from the incense stick) while passing through the Kelvin-Helmholtz instability (rolling vortices).
** This post is part of the ‘Flow series’ by FYPhysics and EcstasyShots!. Check out the previous posts here.
Post link
Flow galleria (#1) : Oil film viewed using Polarizing Filters
In this image a film of oil placed in between plastic sheets is captured by passing the background light through two polarizing filters.
** This post is part of the ‘Flow series’ by FYPhysics and EcstasyShots!. Check out the previous posts here.
Post link

When you wake up in the morning and open/close your faucet when you brush your teeth, you might have noticed that it undergoes a transition between a smooth jet to a dripping flow like so :

When the velocity of the fluid exiting the faucet is high, it appears smooth for a longer time before it breaks into droplets:

But when you make the velocity of the fluid exiting the faucet low, it seems to form droplets much earlier than before.
Here’s the water breaking into smaller droplets shot in slow motion:

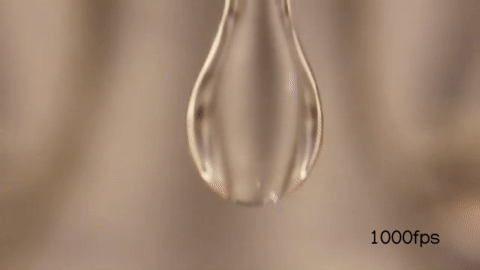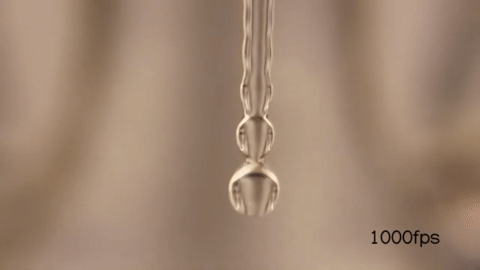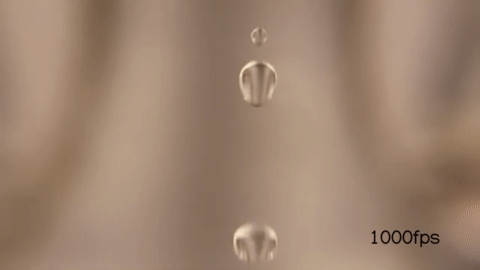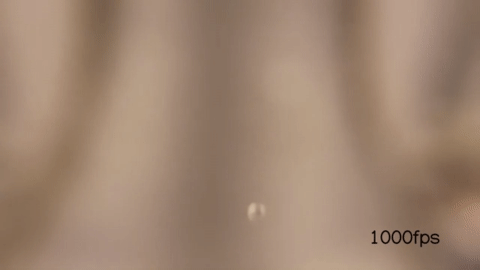
Notice that just by changing the exit velocity of the water you can control when the droplets form.
You can also control the nature of the droplets that form by changing fluids. Here’s how it looks like if you use water as the fluid (left), pure glycerol(center) and a polymeric fluid(right).

What is causing a jet of fluid to form droplets?
A simple answer to this is perturbations on the surface of the fluid. What does that mean?
Initially the fluid is just falling under the influence of gravity. And velocity of any freely falling object increases as it falls:

But the surface tension of the fluid holds the molecules of the fluid together as they fall down.
Therefore depending on the initial velocity of fluid, the surface tension of the fluid and the acceleration you get a characteristic shape of the jet as it falls down:

This is what you observe as the fluid exits the faucet.
Perturbations
Just after exiting the faucet, there are tiny perturbations on the surface of this fluid as it falls down. This is apparent when you record the flow at 3000fps:

Source: engineerguy
Those tiny perturbations on the surface of the fluid grow as the fluid falls down i.e the jet becomes unstable.
And as a result the fluid jet breaks down to smaller droplets to reach a more thermodynamically favorable state. This is known as the Plateau-Rayleigh instability.

It takes different fluids different time scales to reach this instability. This depends on the velocity of the fluid, the surface tension and the acceleration it experiences.

And some viscous fluids like honey are also able to dampen out these perturbations that occur on their surface enabling them to remain as fluid thread for an extended time.
A note on inkjet printers
By externally perturbing the fluid instead of making the fluid do its own thing, you can make droplets of specific sizes and shapes.
This engineerguy video explains how this is used in inkjet printers in grand detail. Do check it out.
We started today by trying to understand why water exiting a faucet behaves the way it does. Hopefully this blog post has gotten you a step closer to realizing that. Have a great day!
Sources and more:
* This is a topic that is home to a lot of research work and interesting fluid dynamics. If you like to explore more take a look into the mathematical treatment of this instability here.
** This post is part of the ‘Flow series’ by FYPhysics and EcstasyShots!. Check out the previous posts here.

Flow(#2) - Plateau- Rayleigh Instability
In this week on FYPhysics! we will be talking about the classic Plateau-Rayleigh Instability. Stay tuned!
** This post is part of the ‘Flow series’ by FYPhysics and EcstasyShots!. Check out the previous posts here.

One of the most surprising things about air that may not be intuitive is that it is a fluid and like any other fluids exerts a pressure on objects.
Standing on earth with layers of air above us, we are constantly being ‘weighed down’ by a pressure of ~1atm at all times.
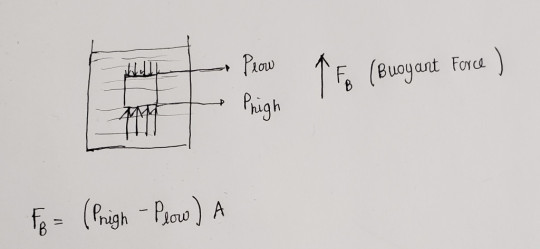
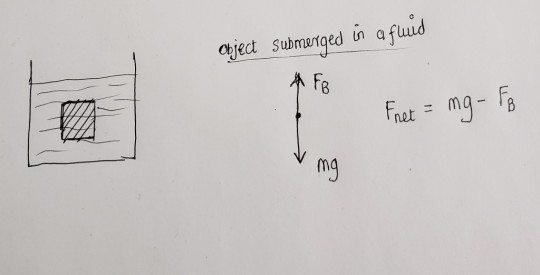
All objects in air are also assisted by a buoyant force that is caused from the pressure difference between the top and bottom surfaces.
Let’s now consider the hot-air balloon in particular. A force diagram is probably the best way to start:

Therefore we see that in order for the hot air balloon to float, we need to have the buoyant force compensate for the net weight from the balloon, the load on it and the air inside it:
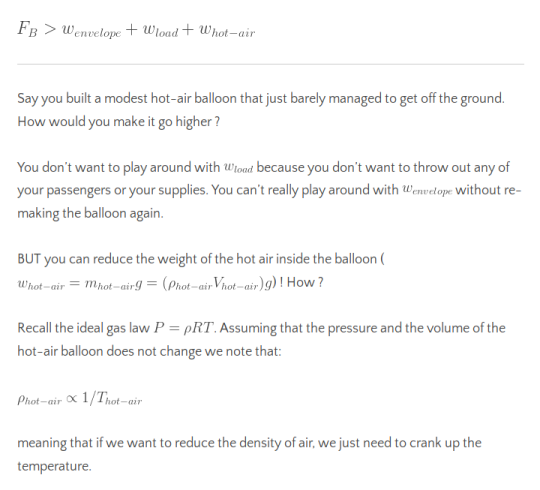

And with a spectacular burner onboard our hot air balloon, we can easily increase the temperature of the air inside the balloon!
That’s pretty much how hot-air balloons work! You increase the temperature of the air inside the balloon to go up, decrease the temperature to go down and skillfully adjust the temperature to hover.
Have a great day!
Questions to ponder:
- When you increase the temperature of air, its density decreases. What do you think happens to all the molecules that were previously inside ? Do they exit the balloon ?
- Different shaped hot-air balloons is a common sight. Do you think that the Buoyant force changes for each shape?
- How do you think Helium balloons work ?
Buoyancy
In the first post of our fluid mechanics series : ‘Flow’ we explore the concept of Buoyancy. Click here to check out the post exploring the physics of Buoyancy.
Have a great day!
* Check out the video description on YouTube on the details of how buoyancy is related to each clip in the video
Flow
Premiering in the summer of 2019 is a web series that we have been working on titled ‘Flow’ which is aimed at expressing the beauty of Fluids through the looking glass of a slow motion camera.
Here is a trailer to the upcoming series. Hope you guys enjoy it!
–
FYPhysics! & Ecstasy Shots!
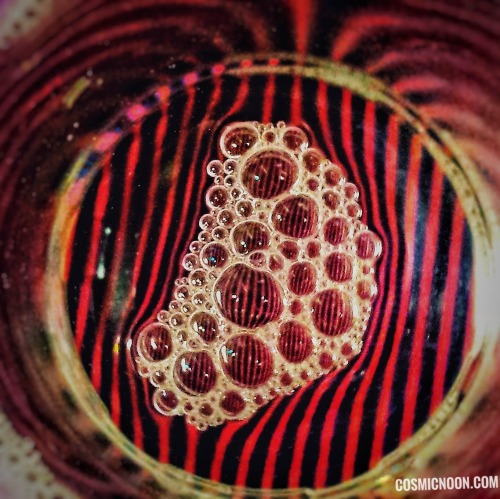
Flow Galleria (#6) : Magic of a bright striped background
One of the easiest way to visualize fluid phenomenon is by placing your fluid on top of a bright striped background. And by measuring the deviations of this striped pattern, it is possible to infer a lot of sensible data such as the changes in density of the fluid and so on. In this post, this technique is demonstrated in water using an alternating red-black striped pattern as the background.
If the region of interest is only the surface on the other hand, the bright striped pattern can be made to reflect off the top of the fluid surface. We demonstrated this technique in our ‘Water is diamagnetic: A simple experiment’ post which you can check out here.
Cheers!
Post link
Flow Gallleria(#3): Cup of water against a striped background
One interesting variation on the cup of water against an arrow in the background (top gif) is if you replace the arrows with a striped background. This opens the door for some interesting visualizations on refraction as well. Try it out!
** This post is part of the ‘Flow series’ by FYPhysics and EcstasyShots!. Check out the previous posts here.
Post link
“Highly magnetizable superparamagnetic colloidal aggregates with narrowed size distribution from ferrofluid emulsion
Volodymyr Lobaz, Robin N. Klupp Taylor, Wolfgang Peukert, ”
Post link
‘Summer with Oki - Free Wallpaper!’
This is a big thanks to all the wonderful followers (even if you’re not, you’re wonderful too!), and I hope you guys have a wonderful summer <3
The high res found on my Patreon for free here: https://www.patreon.com/posts/summer-wallpaper-20802943
——–
If you enjoy my works, please consider supporting me on Patreon <3 | https://www.patreon.com/Werethrope
Post link

Or just quietly do it when your patient’s back is turned…
Suction canisters full of phlegm, anyone? x_x

![Stone Ocean Villain Run 4: [White Snake]Say your sweetest prayer and hope the right God answers youW Stone Ocean Villain Run 4: [White Snake]Say your sweetest prayer and hope the right God answers youW](https://64.media.tumblr.com/ae7ba44fd554fd5cc06a05603b215973/30aff6a33c591ab7-7c/s500x750/ac487b94c478011735546915dbff617873ab8403.png)