#neurology
A Genetic Answer to the Alzheimer’s Riddle?
What if we could pinpoint a hereditary cause for Alzheimer’s, and intervene to reduce the risk of the disease? We may be closer to that goal, thanks to a team at the University of Kentucky. Researchers affiliated with the UK Sanders-Brown Center on Aging have completed new work in Alzheimer’s genetics; the research is detailed in a paper published today in the Journal of Neuroscience.
Emerging evidence indicates that, much like in the case of high cholesterol, some Alzheimer’s disease risk is inherited while the remainder is environmental. Family and twin studies suggest that about 70 percent of total Alzheimer’s risk is hereditary.
Recently published studies identified several variations in DNA sequence that each modify Alzheimer’s risk. In their work, the UK researchers investigated how one of these sequence variations may act. They found that a “protective” genetic variation near a gene called CD33 correlated strongly with how the CD33 mRNA was assembled in the human brain. The authors found that a form of CD33 that lacked a critical functional domain correlates with reduced risk of Alzheimers disease. CD33 is thought to inhibit clearance of amyloid beta, a hallmark of Alzheimers disease.
The results obtained by the UK scientists indicate that inhibiting CD33 may reduce Alzheimer’s risk. A drug tested for acute myeloid leukemia targets CD33, suggesting the potential for treatments based on CD33 to mitigate the risk for Alzheimer’s disease. Additional studies must be conducted before this treatment approach could be tested in humans.
Post link
There is no evidence that impaired blood flow or blockage in the veins of the neck or head is involved in multiple sclerosis, says a McMaster University study.
The research, published online by PLOS ONE Wednesday, found no evidence of abnormalities in the internal jugular or vertebral veins or in the deep cerebral veins of any of 100 patients with multiple sclerosis (MS) compared with 100 people who had no history of any neurological condition.
The study contradicts a controversial theory that says that MS, a chronic, neurodegenerative and inflammatory disease of the central nervous system, is associated with abnormalities in the drainage of venous blood from the brain. In 2008 Italian researcher Paolo Zamboni said that angioplasty, a blockage clearing procedure, would help MS patients with a condition he called chronic cerebrospinal venous insufficiency (CCSVI). This caused a flood of public response in Canada and elsewhere, with many concerned individuals lobbying for support of the ‘Liberation Treatment’ to clear the veins, as advocated by Zamboni.
“This is the first Canadian study to provide compelling evidence against the involvement of CCSVI in MS,” said principal investigator Ian Rodger, a professor emeritus of medicine in the Michael G. DeGroote School of Medicine. “Our findings bring a much needed perspective to the debate surrounding venous angioplasty for MS patients".
In the study all participants received an ultrasound of deep cerebral veins and neck veins as well as a magnetic resonance imaging (MRI) of the neck veins and brain. Each participant had both examinations performed on the same day. The McMaster research team included a radiologist and two ultrasound technicians who had trained in the Zamboni technique at the Department of Vascular Surgery of the University of Ferrara.
Mapping the consequences of our decisions in the brain
Scientists at the University of Glasgow have shed light on how our brain helps us to make decisions.
Imagine picking wild berries in a forest, when suddenly a swarm of bees flies out from behind a bush. In a split second, your motor system has already reacted to flee the swarm. This is the automatic response - acting before thinking - which helps you to avoid impending doom.
At the same time a separate, more thought-out process of learning happens to avoid reckless berry picking in the future.
To understand the way this happens, Dr Philiastides’ team at Glasgow have developed a new way to study brain activity in real-time.
This method involves using two pieces of equipment simultaneously: an EEG machine which records when brain activity took place and a functional MRI scan to reveal where it occurred.
To successfully record from both pieces of equipment at the same time, the team were able to remove the ‘noise’ introduced by the MR scanner on the EEG - a shortcoming that previously made this problematic.
The team hope that this pioneering technique will offer neurological insights into mental disorders associated with decision making such as obsessive-compulsive disorder and post-traumatic stress disorder.
Image credits: Dr Marios Philiastides, University of Glasgow
Post link
The Pitié-Salpêtrière Hospital was originally a gunpowder factory in the 17th century but then morphed into this beast of a hospital. It became the world’s largest on the eve of the French Revolution.
The painting on the final image (“A Clinical lesson at the Salpetriere”) shows the time when the clinical lead Jean-Martin Charcot made the hospital renowned for neuropsychiatry. Here he is showing a young lady with “hysteria” to his students which included names like Sigmund Freud, Babinski, Janet, Tourette and many others.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
#paris #hysteria #architecture #theparisguru #paris_focus_on #parisphotography #parisestmagique #hysterical #parisgram #parismaville #parislifestyle #pariscartepostale #architectureporn #parisianlife #neuropsychology #historyofmedicine #travelawesome #wanderlust #instatravel #francephoto #igersfrance #parisgram #archaddicts #psychiatry #neurology #parisjetaime #igersparis #parisienne #parisian #medicine #hospital (at 13eme arrondissement, Paris)
https://www.instagram.com/p/B7S0CQ1FmhB/?igshid=drr9fkrrpjsw
Post link

232 of 366
I’m gonna start rotating in the pediatric wards on Monday! I got assigned to Neurology subspecialty so here I am brushing up on some cases I might encounter. I also need to review on neuroanatomy really soon. Wish me luck!
ig:studyingdoc
A breakthrough neuromodulation system rapidly restores motor function in patients with a severe spinal cord injury (SCI), new research shows.
The study demonstrated that an epidural electrical stimulation (EES) system developed specifically for spinal cord injuries enabled three men with complete paralysis to stand, walk, cycle, swim, and move their torso within 1 day.
SCIs involve severed connections between the brain and extremities. To compensate for these lost connections, researchers have investigated stem cell therapy, brain-machine interfaces, and powered exoskeletons.
However, these approaches aren’t yet ready for prime time.
Albert Einstein once said to not let your brain interfere with your heart…. but this doggo is proving that to be pretty difficult right now…
Thanks for the pic and posi vibes, @thegoldenlife!
Post link
A Hospitalglam photoset from my not very useful neurologist appointment. She told me we would probably never know what caused my chronic migraines and told me my headaches were from using pain medication.
My mum (acting as my advocate) gave her what for and it helped.
Post link
Some Neurology HospitalGlam. My neurologist is brilliant. Treats me like an equal and partner, reasons out management and plans of action with me, takes my input at face value, never patronizes me or doubts my symptoms or reports. To top it all off, fantastically and brutally honest. From a previous appointment ‘well, there’s still a lot we don’t know about the brain, so the answer probably lies there somewhere.’ And from today 'it definitely sounds like another TIA but we’re already doing the treatment so there’s not much we’d change.’
I’ve worked damn hard to build my current medical team and they may be inexperienced with connective tissue patients, but they are pretty fine doctors.
Post link
– Days 1, 2 & 3
Here’s a quick update to changes to the treatment plan for this year:
1) The ‘anti-listeria’ diet we are asked to follow has been extended to a month before the treatment starts. We had of course been told this prior to this meeting, but she explained that it was because there had been a death caused by listeria in a patient receiving Alemtuzumab. (Thank goodness I’d followed the instructions, hey!)
2) We would be taking a second tablet (in addition to the acyclovir) home with us this year; it’s an antibiotic called Co-Trimoxazole (2x480mg a day for 30 days, each containing: 80mg Trimethoprim & 400mg Sulfamethoxazole), which has been shown to decrease the risk of getting ill whilst our immune systems are very low.
So, for the second round of Alemtuzumab I had 3 days of treatment. I’m not entirely certain why it’s shorter than the first year, but I do know that this is the standard protocol. This year wasn’t very different from last year except, I suppose, that it was a little easier. I’m not sure if that was because I knew what to expect, or that fewer days meant that my body felt less bombarded (or maybe a bit of both?), but either way, I was glad for it!
(This is Morriston the sloth (named after the hospital that my neuro team are based in) sitting in the chair that was my home for the three days of my treatment this year.)
Day one
We started today with some introductions: Mark & I had a new buddy this year called Jenny (also in her 2nd year) and we all met the new neurologist that has been added to our neuro team; both were great. We then each had a little consultation with said neuro who just went through a few things and did a quick series of tests. All we had to do then was wait for the results of our urine tests, take an antiviral & antihistamine, to be cannulated and we were good to go.
There was around an hour of steroids and then 4ish hours of the Alemtuzumab, followed by 2 hours of observation and a blood test. We had half-hourly obs all day and we mainly looked after by 2 nurses. The unit we were in was being shared with the renal department so there was a steady stream of their patients coming in and out for various treatments all day.
I spent this day in quite a lot of discomfort but I knew it was to be expected. The nurses gave me some paracetamol and said that it was good because it meant it was working. At the time all I wanted to do was cry, but looking back on it that was the worst part of the treatment this year and it really wasn’t that bad.
Mum & I stayed in a hotel again as it was much easier and safer to be near the hospital. I was pretty tired after the first day but when trying to sleep I became VERY restless.
(Cannula number 1)
Day two
I had kept the cannula in from the day before so today started a lot faster than the first. All I had to do was wait for my urine test to show all clear and for the blood results from day one to confirm that the treatment had done what it was meant to do. Both were fine, so, after taking an antiviral and antihistamine again, off we went. I had a headache this day and was quite sleepy, but that was pretty much it.
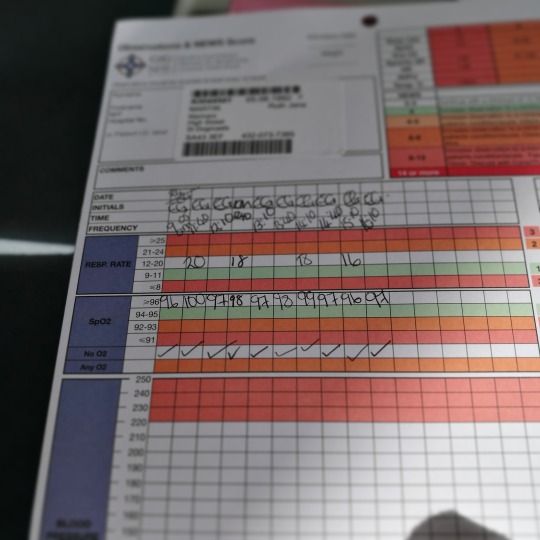
(Half of the obs sheet from day 2)
Day three
I’d kept the cannula in again for this day but about 30 minutes into the steroids it had to be taken out and replaced on the other side. It was a little painful and the infusion seemed to be going quite slowly so it was decided that it was best to change it. Turned out that was a good idea as when it came out the tube was so bent they didn’t even know how it had been going through in the first place (haha). Apart from that the same morning routine from the day before was followed. I was a little flushed this day and had a bit of a temperature but they both went down by the end of the day. I was a lot more tired so spent a lot of the day in and out of sleeping. I did get a small rash, but NOTHING compared to the one I got last year!
So, that was Round 2…hopefully my last! I have a check-up appointment with one of my neurologists in a month and will have an MRI in June(ish) and then we’ll go from there. Keep your fingers crossed for me!
I just wanted to add a quick thank you to my Mum, sisters and friends that looked after me and messaged me throughout these 3 days! Oh, and the awesome neuro team at Morriston (thank goodness for the NHS!!)!
I haven’t had to update this in a while, which has been good, in this case no news really has been good(ish) news.
So what’s happened since last April? To sum up:
> I caught so many bugs it was unbelievable, but none of them caused any hospital visits and as my immune system regained its strength I did stop catching everything.
> I have Trigeminal Neuralgia & take Carbamazapine to stop it from causing me pain, which coincidentally has also seemed to help my back problems.
> I still have to use crutches every so often, but much less than I was starting to use them.
> I’ve had 3 mysterious skin allergic reactions (as shown in previous post), around 10 issues with my eye - itching/pain - that was stopped by piriton - & now have very dry skin/eczema on my eyelids (Aveeno cream clears it up within 24 each time). The GP I saw has suggested that this is all an allergic reaction to the TN meds, but the TN is definitely still worse than all of that. I’m yet to see if my neuro agrees…
> I had a 2 week spell of headaches behind the eyes couple with some mild vertigo recently, but optician couldn’t find an issue, neither could the GP. Again, yet to really bring it up with anyone on the MS team.
> Fatigue, stiffness and memory issues are unfortunately still ever present, but I think they might be something that stick with me for ever to be honest.
> There have (of course) been other issues here & there, but right now none spring to mind…
First things first, my 6 month lymphocyte count is 0.9 - nearly within ‘normal’ range according to my neurologist and is definitely a good thing.
My appointment gave me the opportunity to bring up a few things I’d been worried about/experiencing. Nothing too bad, just a few things that may need more attention next year.
I was also able to catch up briefly with Mark (we had Lemtrada at the same time) which was good. He’s doing well too. We both have to have the Flu jab this year - something I’m definitely not looking forward to.
I’m now taking 50 mg of Carbamazapine twice daily to help with the trigeminal neuralgia - something which my neuro said was a MS thing, but when someone with MS has it, it’s a bit different (it’s caused by the demyelination). It’s being caused by one of my original lesions, so nothing to worrying about on that end (in other words: it’s not new).
Serotonin map of brain could lead to better targeted antidepressants
Researchers hope to discover how the activity of serotonin in the brain is involved in different mental illnesses
Research that aims to map the activity of serotonin in the brain could revolutionise the use of antidepressants and behavioural therapy for people with mental illnesses.
The neurotransmitter serotonin has long been associated with mood, with drugs that boost the chemical in the brain helping to alleviate the symptoms of common illnesses such as depression and anxiety, but scientists lack a deep understanding of how it mediates different mood disorders.
Post link
Splashing neurons with colour to track their development.
A big problem when trying to monitor the ways in which cells change over time is that of consistently identifying the same one. In the tangled billions of neurons in animal brains, this careful art only becomes more complicated.
A research team headed by the University of Southampton has described a possible solution to this problem: use a viral vector to uniquely colour individual cells to keep track of them. Pretty much any RGB colour can be chosen to paint cells as is needed. This way, the problem is reduced to a matter of finding the ‘red one’ among the rest.
The colouration of cells marked in this way is permanently ingrained in DNA, and so is inherited by any daughter cells produced. Using this technique, scientists may soon better understand how connections are made in the brain, how early embryonic development occurs and more simply by watching those processes at the cellular level.
Post link
Fighting Multiple Sclerosis With Cold
Cold exposure therapies could help alleviate symptoms of multiple sclerosis by depriving the immune system of its energy.
Post link
Burning and Tingling in Your Feet? You May Have Small Fiber Neuropathy
Do you experience frequent episodes of burning or tingling in your feet? You may be suffering from small fiber neuropathy, researchers report.
Post link
Does Parkinson’s Start in the Nose?
A majority of Parkinson’s patients report a diminished sense of smell, which starts to occur a number of years before the onset of other symptoms. Researchers are exploring whether scent-processing neurons which connect the nose to the brain may play a role in the development of Parkinson’s disease.
Post link
“THE POWER OF MUSIC” TO AFFECT THE BRAIN
“…scientists have found that music stimulates more parts of the brain than any other human function. That’s why she sees so much potential in music’s power to change the brain and affect the way it works.”
Read:“The Power Of Music” To Affect The Brain
Post link
[The] UC San Diego-led team will receive a $9 million grant from the Aligning Science Across Parkinson’s (ASAP) initiative to help advance this research and position it for the next phases of drug development. ASAP is a coordinated research initiative to advance targeted basic research for Parkinson’s disease. Its mission is to accelerate the pace of discovery and inform the path to a cure through collaboration, research-enabling resources and data-sharing. The Michael J. Fox Foundation for Parkinson’s Research is the implementation partner for ASAP and issuer of the grant, which contributes to the Campaign for UC San Diego.
“This grant is supporting some of the most incredible progress being made in the Parkinson’s sphere. It’s a game-changing strategy that we hope will improve how Parkinson’s is treated,” said David Brenner, MD, vice chancellor of Health Sciences. “We are grateful to ASAP for making these advancements possible.”
This post on visual snow had me realizing that there was a nameto the blood-colored static i tried to explain to a friend a while back while comparing how accurately we could hold an image in our heads. Up until a couple of years ago, i had thought that mental imagery was roughly the same for everyone other than those with eidetic memory, and was baffled to learn that some people could barely hold images in their mind at all (some of those people are still able to draw for a living???)
At the time i had created some visuals to attempt to explain how my thoughts ‘looked,’ and since i still think they’re interesting i figured i’d post them.